Video: The 9 Hallmarks of Aging, episode 3, Epigenetics – Guilhem Velvé Casquillas, PhD
This time we will address a third identified cause of aging: epigenetic changes.
Epigenetics – Definition
But first, what is epigenetics? We all know genetics well, the science that looks at DNA, the information it contains and the alterations of that information by mutations in the genes. Epigenetics, on the other hand, looks at all the mechanisms that will allow genes to be expressed and how these genes will be used without modifying our DNA sequences. Epigenetics therefore highlights the variability of the expression of a gene as a function of time and its environment. A simple way to understand the strength of epigenetics is to look at the organs: each cell in your body contains the same genetic information, and yet, depending on their physico-chemical environment, some cells will give neurons, skin cells or heart muscle cells. A good example of the strength of epigenetics (you can find it on wikipedia), is with bees. A larva can become a queen or a worker who lives ten times less long depending on how she is fed. It is a perfect example of the epigenetic phenomenon: the same information at the beginning for a different result.
This area is very interesting for understanding aging and we have identified 3 main epigenetic modes of action that can influence our lifespan: DNA methylation, histone modification and chromatin remodeling.
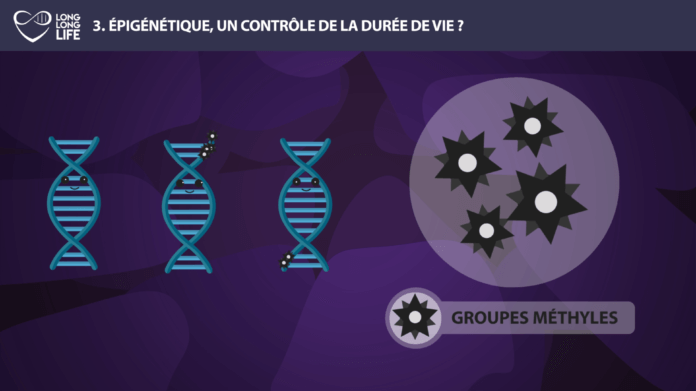
Epigenetics – Methylation
Methylation is an epigenetic phenomenon that will allow the addition or removal of a small molecule, called methyl, on certain parts of our DNA. These changes can alter the expression of one gene, increasing the production of one protein or decreasing the synthesis of another. This methylation can affect many genes with a more or less important role depending on the role of the affected gene. Over time, the epigenetic changes associated with methylation increase and this could be the indirect cause of problems, such as increased cholesterol levels, or cardiovascular risks.

And it’s the same with histones! Histones are the proteins around which DNA is wound to form chromosomes. Like DNA, these histones can be modified by adding or removing small molecules. This time, it is the acetyls (and no longer the methyl) that interest us: in invertebrates, we observed that the suppression of these acetyls on a specific histone could increase their life expectancy. These changes can therefore have a significant effect on the body and are able to modify the expression of a gene.
Epigenetics – Chromatin
Now it’s chromatin’s turn. Chromatin is the form in which DNA is found in the nucleus. It allows DNA packaging to store maximum information in tiny spaces. This compaction takes place in particular thanks to the histones we have just mentioned. But chromatin can also be remodelled, i.e. it will be more or less condensed. In its very condensed form, it will be called heterochromatin and in its less dense form, it will be called euchromatin. With age, chromatin remodelling decreases, the balance between heterochromatin and euchromatin is no longer respected and this affects not only chromosome stability but also gene expression. Studies have shown that chromatin regulation is an important factor in telomere length control. Thus this epigenetic alteration can lead to accelerated aging through telomere shortening.

In summary, the study of epigenetic alterations is at the heart of research on aging. DNA methylation, histone modification and chromatin remodelling are altered processes over time and have been shown to be linked to age-related diseases such as cancer, atherosclerosis and dementia.
Our articles on the epigenetics of aging:
Dr Guilhem Velvé Casquillas

Author/Reviewer
Auteur/Relecteur
Physics PhD, CEO NBIC Valley, CEO Long Long Life, CEO Elvesys Microfluidic Innovation Center
More about the Long Long Life team
Docteur en physique, CEO NBIC Valley, CEO Long Long Life, CEO Elvesys Microfluidic Innovation Center
En savoir plus sur l’équipe de Long Long Life






![[Video] The 9 Hallmarks of Aging, Episode 9, Inflammation [FINAL] – with Guilhem Velvé Casquillas PhD](https://www.longlonglife.org/wp-content/uploads/2019/05/Inflammation-Long-Long-Life-longévité-vieillissement-transhumisme-inflammaging-1-218x150.png)
![[Video] The 9 Causes of Aging, Episode 8, Stem Cells – with Dr. Guilhem Velvé Casquillas cellules souches long long life transhumanisme longévité vieillissement arcade](https://www.longlonglife.org/wp-content/uploads/2019/05/cellules-souches-long-long-life-transhumanisme-longévité-vieillissement-arcade-218x150.png)
![[Video] – The 9 Causes of Aging, Episode 7, Senescence – with Dr. Guilhem Velvé Casquillas sénescence long long life longévité transhumanisme vieillissement cellules sénescentes](https://www.longlonglife.org/wp-content/uploads/2019/05/sénescence-long-long-life-longévité-transhumanisme-vieillissement-cellules-sénescentes-1-218x150.png)
![[Video] – The 9 Causes of Aging, Episode 6, Nutrient Detection – with Dr. Guilhem Velvé Casquillas nutriments 1 long long life transhumanisme longévité vieillissement](https://www.longlonglife.org/wp-content/uploads/2019/05/nutriments-1-long-long-life-transhumanisme-longévité-vieillissement-218x150.png)
![[Video] The 9 Hallmarks of Aging, Episode 5, Mitochondria – with Dr. Guilhem Velvé Casquillas mitochondria long long life longevity transhumanism aging free radicals (2)](https://www.longlonglife.org/wp-content/uploads/2019/07/mitochondria-long-long-life-longevity-transhumanism-aging-free-radicals-2-218x150.png)
![[Video] Eurosymposium on Healthy Ageing, Brussels, 2018 Eurosymposium on Healthy Aging](https://www.longlonglife.org/wp-content/uploads/2019/07/P1310252-218x150.jpg)
![[Video] The 9 Causes of Aging, Episode 4, Poorly Folded Proteins – with Dr. Guilhem Velvé Casquillas protéine long long life 9 causes du vieillissement longévité transhumanisme](https://www.longlonglife.org/wp-content/uploads/2019/05/protéine-long-long-life-9-causes-du-vieillissement-longévité-transhumanisme-218x150.png)
![[Video] The 9 Hallmarks of Aging, Episode 2, Telomere shortening – Guilhem Velvé Casquillas, PhD Télomère - Long Long Life 9 causes du vieillissement longévité transhumanisme](https://www.longlonglife.org/wp-content/uploads/2019/06/Télomère-Long-Long-Life-9-causes-du-vieillissement-longévité-transhumanisme-1-218x150.png)



![[Video] – The 9 Causes of Aging, Episode 6, Nutrient Detection – with Dr. Guilhem Velvé Casquillas nutriments 1 long long life transhumanisme longévité vieillissement](https://www.longlonglife.org/wp-content/uploads/2019/05/nutriments-1-long-long-life-transhumanisme-longévité-vieillissement-324x160.png)