[Video] The 9 Hallmarks of Aging, Episode 2, Telomere shortening
We will address a second identified cause of aging, the shortening of telomeres, which are often considered to be the biological clock of our cells.
Telomere shortening – Definition
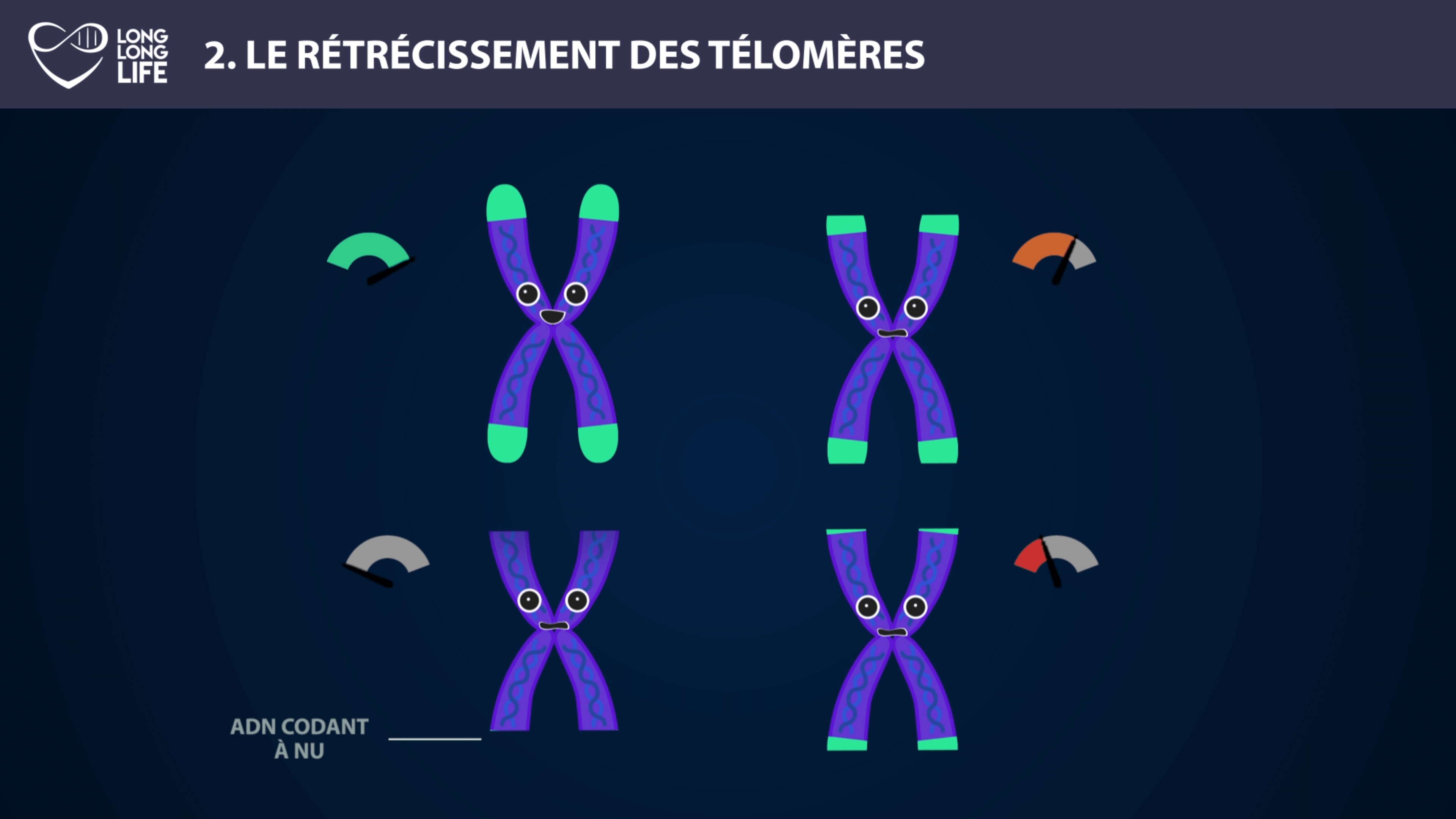
What are telomeres? At each of the four ends of the chromosome is a small DNA cap called a telomere that protects the chromosome. Unfortunately, they are nibbled at each cell division, until they disappear and leave the DNA bare. When this happens, the DNA is no longer able to replicate itself without making mistakes and this causes the cell to die.
So what’s going on? When a cell divides, it replicates its DNA to transfer genetic information to the new cells it created. In fact, with each cell division, a molecule called DNA polymerase duplicates our genetic code. But DNA polymerase “forgets” to reproduce the end of the DNA structure, in this case telomeres.
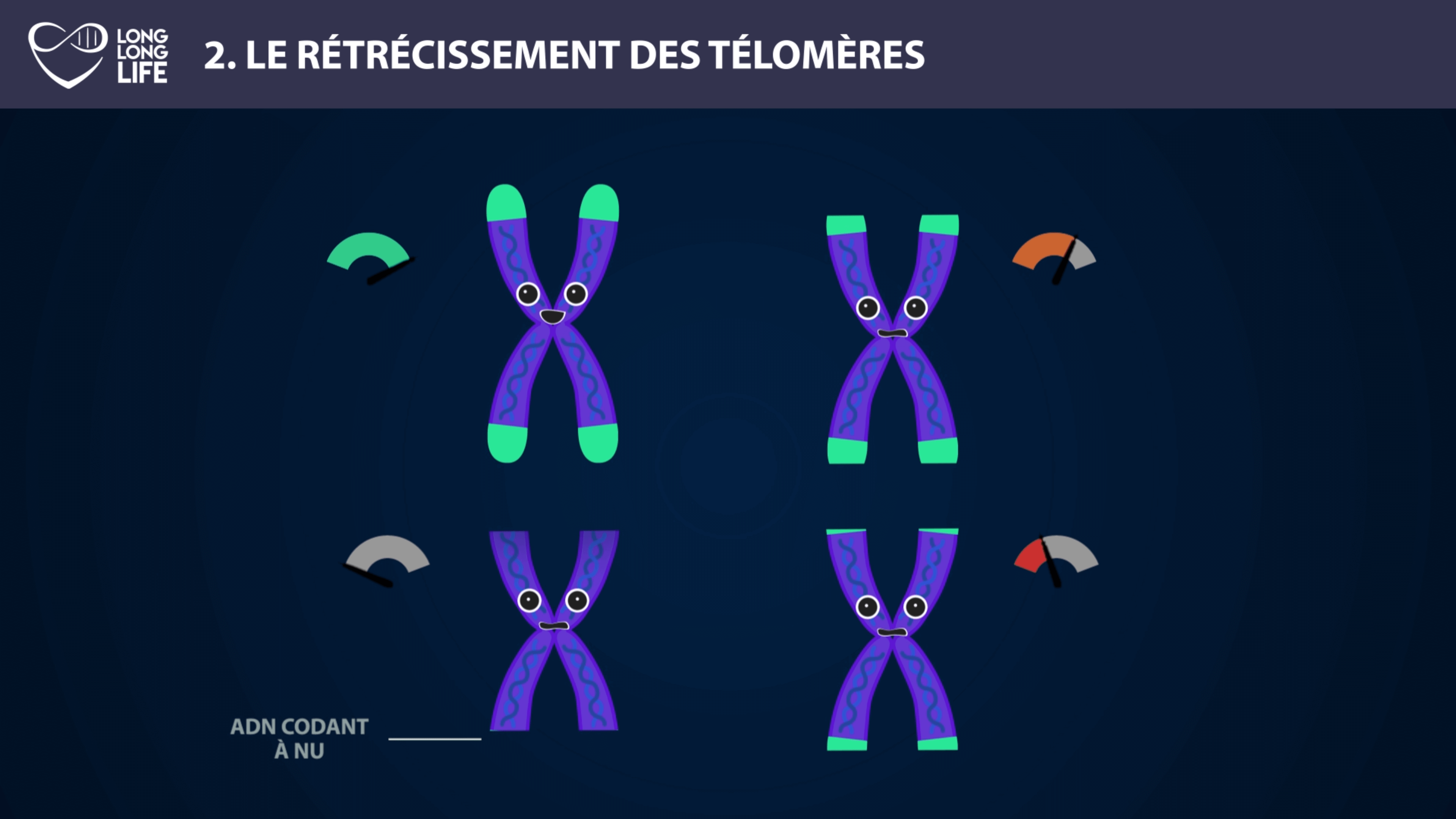

Telomere shortening – Telomerase
This function is performed exclusively by a special enzyme known as telomerase. But it would be too good for all our cells to express telomerase, they would be almost immortal! This enzyme is present only in certain cells. First of all in our stem cells, the cells capable of creating all our specialized cells, liver cells, muscle cells, fat cells…

Telomerase is also present in our so-called “germ cells”, i.e. cells allowing our reproduction, and finally it is also present in a few classic cells called “somatic”, such as hair follicle cells. For the others, it is a programmed death that awaits them, defined by Hayflick’s limit. Dr Hayflick discovered that there were a maximum number of divisions that a cell could carry out… and that this number was dependent on how long the telomeres were. Indeed, in the absence of telomerase, the cells are not able to reconstruct their telomeres and the cell will decide to die rather than to replicate its DNA with missing information.
Unfortunately for us, another type of cell has telomerase, it is cancer cells. 90% of these cells express telomerase, which makes them almost immortal and capable of infinite division. And these cancer cells highlight the dilemma of telomerase: their shortening causes our cells to die and our tissues to age, but protects us from cancer.
Telomere shortening – Current research
So, as you can imagine, some researchers have tried to evaluate the possibility of using telomerase to rejuvenate our tissues.
On old mice, a Harvard team has activated the genes responsible for telomerase production. These experiments gave good results, with a rejuvenation of mice and a good regeneration of damaged tissues.

Another study was conducted on skin cells. In culture these cells can divide between 40 and 60 times during their life. By genetically modifying them to activate telomerase production, these same cells were able to divide up to 300 times.
These two experiments show the importance of telomeres in the life expectancy of a cell but the associated risks, such as cancer development, are well present and no studies have been conducted in humans to date.
This did not stop Liz Parrish, CEO of Bioviva, from carrying out an experimental gene therapy on herself in 2106 to activate her telomerase expression gene. For the moment the results are encouraging, because she says she has rejuvenated her telomeres by 20 years. But it remains for the moment difficult to conclude on the reality of an increase in her life expectancy, which I wish her with all my heart.
Moreover, for those interested in aging metrology, telomeres can be used as a partial measure of your physiological age and some companies or clinics offer to measure the size of your telomere on samples of your cells.

To sum it up: telomere narrowing can be seen as a biological clock in each cell. This mechanism is a limit to the life expectancy of our cells, which is why it has a very important place in current research against aging.
Curious about telomeres? Find out more with our in-depth articles:
See all of our articles on “Telomeres and aging”:
Telomeres, at the heart of the aging process
 How and why does telomere shortening seem central to the aging process ? What is their role? How do they influence the aging process? And why do we call telomeres the “biological clocks” of our body?
How and why does telomere shortening seem central to the aging process ? What is their role? How do they influence the aging process? And why do we call telomeres the “biological clocks” of our body?
Part 1: Causes and consequences of telomere shortening during aging
 It is unclear how fast and why telomere shortening speed and aging vary from person to person. Indeed, the possible causes of telomere shortening can vary greatly.
It is unclear how fast and why telomere shortening speed and aging vary from person to person. Indeed, the possible causes of telomere shortening can vary greatly.
Part 2: Accelerated aging due to telomere and telomerase malfunction
 Telomere length and telomerase seem to be key factors of the aging process. Many studies on diseases resulting from mutations on telomerase components have shown that it leads to a lesser quality of cell renewal, which is a phenotype linked to aging.
Telomere length and telomerase seem to be key factors of the aging process. Many studies on diseases resulting from mutations on telomerase components have shown that it leads to a lesser quality of cell renewal, which is a phenotype linked to aging.
Part 3: Telomeres and telomerase in stem cells: central to the aging process
 Telomerase expression diminishes in the few weeks that follow birth in most adult tissues, with the exception of certain types of cells such as stem cells. One can wonder if there is a link between the fact that the quantity of stem cells is lowered with age, telomerase function, and telomere length.
Telomerase expression diminishes in the few weeks that follow birth in most adult tissues, with the exception of certain types of cells such as stem cells. One can wonder if there is a link between the fact that the quantity of stem cells is lowered with age, telomerase function, and telomere length.
Part 4: Towards an aging metrology with telomeres
 To measure aging, several methods based on telomere length have been developed. Today, there are 5 main methods, among which TAT, and STELA. They allow to obtain precious indications on physiological age and aging, from telomere length.
To measure aging, several methods based on telomere length have been developed. Today, there are 5 main methods, among which TAT, and STELA. They allow to obtain precious indications on physiological age and aging, from telomere length.
Part 5: Telomeres and aging, what therapies?
 Telomere length is becoming an interesting lead to elaborate therapies and solutions to fight aging. We can speak of Elizabeth Parrish, CEO of BioViva, a biotech company in the US. She tested on herself two gene therapies developed by her own lab, one of which aims to lengthen her telomeres for “rejuvenation” purposes
Telomere length is becoming an interesting lead to elaborate therapies and solutions to fight aging. We can speak of Elizabeth Parrish, CEO of BioViva, a biotech company in the US. She tested on herself two gene therapies developed by her own lab, one of which aims to lengthen her telomeres for “rejuvenation” purposes
Dr Guilhem Velvé Casquillas

Author/Reviewer
Auteur/Relecteur
Physics PhD, CEO NBIC Valley, CEO Long Long Life, CEO Elvesys Microfluidic Innovation Center
More about the Long Long Life team
Docteur en physique, CEO NBIC Valley, CEO Long Long Life, CEO Elvesys Microfluidic Innovation Center
En savoir plus sur l’équipe de Long Long Life


![[Video] The 9 Hallmarks of Aging, Episode 9, Inflammation [FINAL] – with Guilhem Velvé Casquillas PhD](https://www.longlonglife.org/wp-content/uploads/2019/05/Inflammation-Long-Long-Life-longévité-vieillissement-transhumisme-inflammaging-1-218x150.png)
![[Video] The 9 Causes of Aging, Episode 8, Stem Cells – with Dr. Guilhem Velvé Casquillas cellules souches long long life transhumanisme longévité vieillissement arcade](https://www.longlonglife.org/wp-content/uploads/2019/05/cellules-souches-long-long-life-transhumanisme-longévité-vieillissement-arcade-218x150.png)
![[Video] – The 9 Causes of Aging, Episode 7, Senescence – with Dr. Guilhem Velvé Casquillas sénescence long long life longévité transhumanisme vieillissement cellules sénescentes](https://www.longlonglife.org/wp-content/uploads/2019/05/sénescence-long-long-life-longévité-transhumanisme-vieillissement-cellules-sénescentes-1-218x150.png)
![[Video] – The 9 Causes of Aging, Episode 6, Nutrient Detection – with Dr. Guilhem Velvé Casquillas nutriments 1 long long life transhumanisme longévité vieillissement](https://www.longlonglife.org/wp-content/uploads/2019/05/nutriments-1-long-long-life-transhumanisme-longévité-vieillissement-218x150.png)
![[Video] The 9 Hallmarks of Aging, Episode 5, Mitochondria – with Dr. Guilhem Velvé Casquillas mitochondria long long life longevity transhumanism aging free radicals (2)](https://www.longlonglife.org/wp-content/uploads/2019/07/mitochondria-long-long-life-longevity-transhumanism-aging-free-radicals-2-218x150.png)
![[Video] Eurosymposium on Healthy Ageing, Brussels, 2018 Eurosymposium on Healthy Aging](https://www.longlonglife.org/wp-content/uploads/2019/07/P1310252-218x150.jpg)
![[Video] The 9 Causes of Aging, Episode 4, Poorly Folded Proteins – with Dr. Guilhem Velvé Casquillas protéine long long life 9 causes du vieillissement longévité transhumanisme](https://www.longlonglife.org/wp-content/uploads/2019/05/protéine-long-long-life-9-causes-du-vieillissement-longévité-transhumanisme-218x150.png)



![[Video] The 9 Causes of Aging, Episode 4, Poorly Folded Proteins – with Dr. Guilhem Velvé Casquillas protéine long long life 9 causes du vieillissement longévité transhumanisme](https://www.longlonglife.org/wp-content/uploads/2019/05/protéine-long-long-life-9-causes-du-vieillissement-longévité-transhumanisme-324x160.png)
![[Video] The 9 Causes of Aging, Episode 8, Stem Cells – with Dr. Guilhem Velvé Casquillas cellules souches long long life transhumanisme longévité vieillissement arcade](https://www.longlonglife.org/wp-content/uploads/2019/05/cellules-souches-long-long-life-transhumanisme-longévité-vieillissement-arcade-324x160.png)