Transhumanism: Aubrey de Grey’s causes of aging and how to stop them

Dr. Aubrey de Grey is one of the leading figures in anti-aging research. He is the co-founder of SENS, a California-based research institute dedicated to anti-aging research with a strong focus on regenerative medicine. In 1999 he published a book entitled “The Mitochondrial Free Radical Theory of Aging”, in which he explains that the damage inflicted on mitochondria during our lives is the main reason why we age. He then created a scientific journal, Rejuvenation Research, dedicated to the publication of research in the field of aging. Finally, he is himself involved in this research, as a bioinformatician, and regularly participates in conferences, more or less general public. His TED conference was a great success. More recently, he co-wrote a new book “Ending Aging”, even more extensive than his first book.
Aubrey de Grey’s goal? To slow down aging by replacing or editing our cells.
Gerontology, geriatrics and engineering interventions
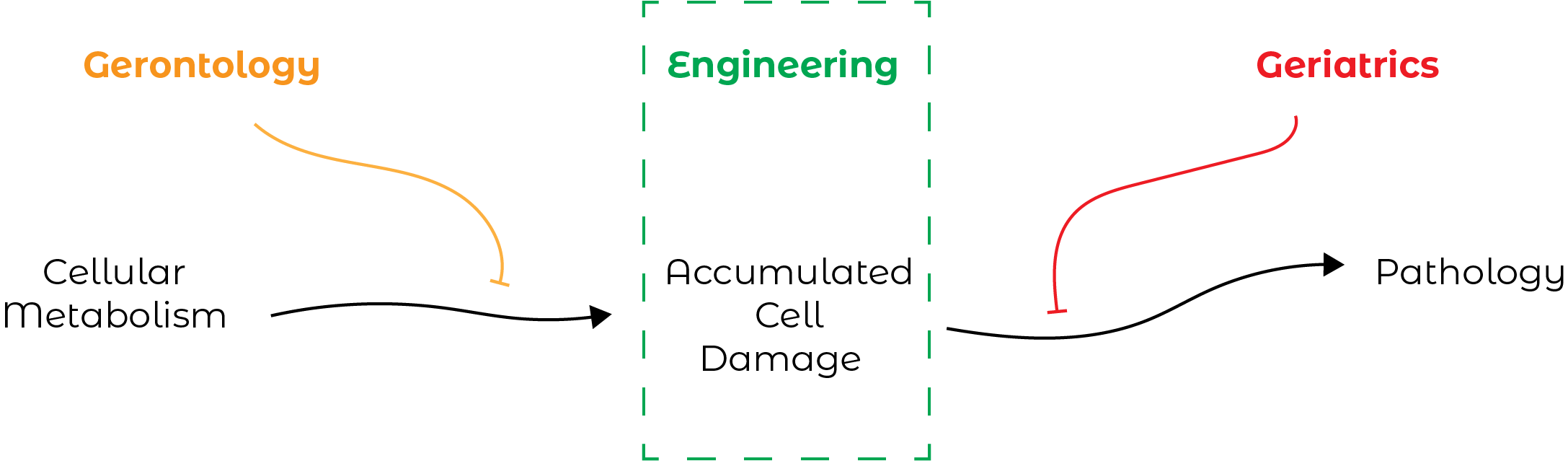
According to Aubrey de Grey, aging can be defined very simply. We have a cellular metabolism, which corresponds to everything allowing us to live from one day to the next and makes our cells function. This metabolism accumulates, over time, damage, intrinsically related to its operation which is not without errors. This damage, when it accumulates, causes pathologies and causes us to age.
Aubrey de Grey’s theories are essentially based on one observation: current approaches to gerontology and geriatrics are not sufficient or sufficiently developed tools to effectively slow aging. He explains that the gerontological approach, which consists of intervening upstream of the cellular damage that takes place during our lives, does not today have tools powerful enough to be effective. This lack of tools is accompanied by a lack of overall knowledge of our metabolism. On the other hand, the geriatric approach, which intervenes after the onset of cellular damage and tries to prevent the apparition of pathologies, is, according to him, a race against time, lost in advance.
It is for this reason that it focuses on an engineering approach, the aim of which is not prevention but pure and simple treatment of aging. Indeed, in gerontology, if we are interested in metabolism, the list of signalling pathways, proteins, enzymes… involved in the processes defining it, is infinite! In geriatrics, if we restrict ourselves to preventing certain diseases, others may appear and it is not possible to take into account all existing diseases. Conversely, with Aubrey de Grey’s engineering approach, the fight against aging will focus on cellular damage, the list of which is very limited and has changed very little over the past 100 years.
The seven causes of aging according to Aubrey de Grey
According to Aubrey de Grey, cellular damage can be classified into seven interconnected parts. These seven causes have remained unchanged since the early 1980s, implying that there is little yet to be discovered in this area. This is encouraging, because it limits us to a few well-defined mechanisms, unlike the study of metabolism (gerontological approach) or pathologies (geriatric approach), which are much more complex and still being discovered!
Intracellular waste
Intercellular waste
Nucleus mutations
Mitochondrial mutations
Stem cells loss
Increase in senescent cells
Increase of intercellular protein links
Cellular waste management
When our metabolism works and in order to produce the proteins we need, to allow cell division or to synthesize hormones or metabolites needed for organ function, our cells will also produce waste products that need to be disposed of. As with all metabolic phenomena, these “cell waste” processes will function well at the beginning of life and will weaken over time. The concept of extracellular waste was highlighted in 1907 by Aloïs Alzheimer, who gave his name to the disease he characterized. Indeed, in Alzheimer’s disease, proteins accumulate outside the neurons preventing their connection with other surrounding cells and causing their death (see Alzheimer’s Disease).
The concept of intracellular waste was discovered by Strehler in 1959. According to him, the aging process has four characteristics: it is destructive, it is progressive (and therefore irreversible), it is intrinsic (and this is what interests us) and it is universal. Cellular waste, in addition to accumulating between cells, can also be stored in cells, when “garbage truck” machinery jams. This is particularly what happens in macular degeneration or atherosclerosis.
Genetic mutations
Aubrey de Grey distinguishes two types of mutations according to the target affected, nuclear mutations (Szilard, 1959 and Cutler, 1982) and mitochondrial mutations (Harman, 1972). A genetic mutation is when our information, stored in the form of DNA, is transformed by various mechanisms. Nuclear mutations, i.e. those affecting the DNA present in our nucleus and coding for almost all the proteins in our body, are the ones we know best, because they are common to many pathologies (cancer, diabetes…). Mutations are generally hereditary, but there are also so-called de novo mutations: when cells divide, they must copy all of their DNA to transmit it to their daughter cells, and during this mechanism, errors can appear. Most often, cells carrying serious mutations will commit suicide through programmed cell death (apoptosis) but sometimes they escape this mechanism and survive, dividing in turn and giving daughter cells carrying the mutation.

At the same time, mutations may appear in mitochondrial DNA. Mitochondria are somewhat special cellular organelles: they provide us with energy (the well-known ATP and NAD), allow cellular respiration (and the synthesis of unfortunate oxidants) and are involved in central mechanisms, such as cell death or growth. It is the result of an ancestral fusion (about 2 billion years) between our cells and a bacterium. Its utility was such that, during evolution, human cells fully integrated it and, because of its origin, it has its own DNA, independent of nuclear DNA, which codes for 13 proteins necessary for it to function: it is the mitochondrial genome. Mutations affecting this DNA are mostly known for mitochondrial diseases, multifaceted pathologies, but, by the same process as for nuclear DNA, mutations can appear de novo.
Managing our cell stock
Several types of cells populate our body: short-lived cells, which often renew themselves, long-lived cells, which do not or only slightly renew themselves, virtually immortal stem cells, and senescent cells, “zombies”, cells at the end of their lives, incapable of dividing but not dying either and whose functions degrade. During our lifetime, the stock of stem cells decreases and the number of senescent cells increases. Aubrey de Grey postulates that this imbalance is one of the origins of aging. This theory is based on the work of Brody (1955) and Hayflick (1972), two researchers whose work has not been called into question until now.
Protein interconnections
For our cells to communicate and form cohesive tissues, they must interact with each other. This cohesion is achieved by specific proteins whose number and/or number of bonds they make tend to increase with age (Monnier & Cerami, 1981). Aubrey de Grey explains that this multiplication of intercellular bonds, far from being beneficial, can cause a loss of tissue elasticity and lead to the appearance of diseases, such as arteriosclerosis.
The future of treatments and the concept of longevity escape velocity
Aubrey de Grey’s approach is as follows: we have identified the seven underlying mechanisms of aging and will soon be able to intervene at the time of cellular damage. In the absence of prevention, we will be able to treat, as and when it appears, the aging of our cells.
This is where two concepts come in: “robust human rejuvenation” and “longevity escape velocity“. The robust human rejuvenation consists of a major discovery, allowing for example to add 30 years of life to a person currently 55 years old. This will be the “eureka” moment of research against aging. From this date, new treatments and refinements of the initial discovery will regularly add years of life in addition to the initial 30 years planned. This is what Aubrey de Grey calls longevity escape velocity and which leads to a simple observation: the first person to live 1000 years will be about 20 years younger than the first man to live 150 years, due to the exponential implementation of treatments. He makes this postulate based on scientific developments in other fields and regularly gives the example of aviation: in 1903, the first aircraft is built (this is the fundamental discovery), in 1927, the first solo non-stop flight over the Atlantic takes place, in 1949, the first jet aircraft is launched and in 1969, the first supersonic flights begin. This is what Aubrey de Grey defines as a growing and exponential improvement of technologies: from the major discovery, technological evolution is rapid and will, in his opinion, regularly add years of life.
What about treatment options?
A solution for every problem. Aubrey de Grey focuses on regenerative approaches to anti-aging medicine, i.e. technologies that allow the normal restoration of tissue structure and/or function after damage has occurred.
Issues | Potential therapies |
|---|---|
| Intracellular waste | Transgenic microbial hydrolases: enzymes capable of destroying the waste, which are not present in our bodies |
| Intercellular waste | Stimulation of phagocytosis by our immune system: macrophages in particular, whose role is to eat waste products |
| Nucleus mutations | KO of telomerase and increase in the number of stem cells: the goal is to decrease the number of cell divisions in order to reduce the risk of mutations, while maintaining a normal level of renewal with stem cells |
| Mitochondrial mutations | Allotropic expression of the 13 proteins encoded by mtDNA through the integration of this DNA sequence into nuclear DNA |
| Stem cells loss | Cell therapy using growth factors and stem cell addition |
| Increase in senescent cells | Removal of senescent cells by targeting, mostly by boosting "suicide" genes |
| Increase of intercellular protein links | Enzymes that can break these protein links between the cells |
Some of these therapies are already well advanced and may soon lead to clinical trials. Important discoveries have also been made in recent years, including iPS cells (stem cells that we can control) and genetic editing tools (see CRISPR-cas9). However, as Aubrey de Grey regularly says, we don’t know when the discovery will come that will change everything and quickly lengthen our healthspans and lifespans!
Dr. Marion Tible

Author/Reviewer
Auteure/Relectrice
Marion Tible has a PhD in cellular biology and physiopathology. Formerly a researcher in thematics varying from cardiology to neurodegenerative diseases, she is now part of Long Long Life team and is involved in scientific writing and anti-aging research.
More about the Long Long Life team
Marion Tible est docteur en biologie cellulaire et physiopathologie. Ancienne chercheuse dans des thématiques oscillant de la cardiologie aux maladies neurodégénératives, elle est aujourd’hui impliquée au sein de Long Long Life pour la rédaction scientifique et la recherche contre le vieillissement.
En savoir plus sur l’équipe de Long Long Life
Dr Guilhem Velvé Casquillas

Author/Reviewer
Auteur/Relecteur
Physics PhD, CEO NBIC Valley, CEO Long Long Life, CEO Elvesys Microfluidic Innovation Center
More about the Long Long Life team
Docteur en physique, CEO NBIC Valley, CEO Long Long Life, CEO Elvesys Microfluidic Innovation Center
En savoir plus sur l’équipe de Long Long Life



![[Video] Eurosymposium on Healthy Ageing, Brussels, 2018 Eurosymposium on Healthy Aging](http://www.longlonglife.org/wp-content/uploads/2019/07/P1310252-218x150.jpg)